To the doctor in the emergency department, the case seemed run-of-the-mill. It was July, 2010, and the patient had just returned from Paris, where he’d ordered a tripe dish from a tourist trap near the Notre-Dame Cathedral. He was now feeling wretched: fever, full-body aches and a stomach in revolt.
Traveller’s diarrhea, the doctor concluded. He reassured the patient that a standard course of antibiotics would do the trick.
But Gerry Wright was not a run-of-the-mill patient. At the time, he was director of the Michael G. DeGroote Institute for Infectious Disease Research at McMaster University in Hamilton, and had already spent decades studying the germs that make people sick – and how wily they can be.
“I remember asking the physician, ‘What do you know about antibiotic-resistance patterns in France?’ ” Dr. Wright recalled. “He was like, ‘I don’t know what you’re talking about. This is going to work; it always works.’ ”
The antibiotics did not work. Two days later, Dr. Wright was “nearly comatose” at McMaster University Medical Centre, where he was seen by specialists. He was given a different antibiotic, which had to be taken intravenously, for more than a week. It would take him several more months to fully recover.
Gerry Wright, professor of biochemistry and biomedical sciences, and Michael Cook, Wright Lab research coordinator, inspect a tray of chemical compounds naturally produced by bacteria and other microorganisms at McMaster University.
After his initial treatment failed, Dr. Wright had a suspicion. He obtained a sample of the pathogen coursing through his bloodstream and sequenced its genome. As suspected, he had a strain of salmonella that was impervious to ciprofloxacin, the first antibiotic he was given.
Dr. Wright’s bug, in other words, was drug resistant.
Today, the 62-year-old academic can appreciate the cruel joke of this miserable episode. The germ that threatened his life was part of an alarming and growing threat that he’s devoted his life to combatting: the rise of the superbug.
Superbugs are bacteria and other pathogens that have become resistant to available drugs; collectively they represent one of the gravest public health threats of our time. Sometimes dubbed a “slow-moving pandemic,” antimicrobial resistance (AMR) has been projected to cause 10 million deaths annually by 2050 – more than cancer’s current toll.
In an imagined future without antibiotics, the modern health care system buckles. Everything – from chemotherapy to C-sections – becomes potentially life threatening. Even a simple scratch could prove fatal.
In the mid-20th century, drug companies spent small fortunes combing through soil, where antibiotics are naturally produced by bacteria and fungi. But despite the dire need, the search for novel antibiotics has been largely abandoned by a pharmaceutical industry that’s shifted toward more profitable ventures. Whereas cancer is studied by tens of thousands of scientists, there are only about 3,000 researchers working on AMR, according to a 2024 industry report.
Dr. Wright is one of them. He has spent his career asking twinned questions. How do superbugs develop resistance? And how can we find new ways to kill them?
Over the decades, he and his team at McMaster’s Wright Lab have found several novel antimicrobial compounds, the chemical substances that kill or inhibit the growth of microbes. But none have been developed into a drug.
Last year, however, his lab published a paper with a thrilling announcement: His team had discovered a promising antimicrobial. And then they announced another one. And another. Today, the Wright Lab has two more novel antimicrobials that they’ve written up and submitted for academic publication.
Trays display chemical compounds naturally produced by bacteria and other microorganisms from McMaster University’s natural products library.
It’s a hot streak that’s unusual in the bleak field of antimicrobial drug discovery, which has become a wasteland of failures. And it’s raising eyebrows across the scientific community.
“That is a terrific rate of success,” said Kim Lewis, director of the Antimicrobial Discovery Center at Northeastern University in Boston, and a globally recognized leader in the field. “They know how to place a bet.”
Academic researchers are, in a sense, gamblers. You form a hypothesis, recruit people and resources to your cause, continually beg for funding to keep the lights on – and after staking your entire academic career on something, you may never find what you’re looking for.
In his quest for the next antibiotic, Dr. Wright bet the house on dirt. It’s a wager some skeptics consider the scientific equivalent of tilting at windmills. After all, his strategy is based on a hypothesis most scientists have long abandoned: that with a spoon, and some soil, new antibiotics can still be found.
But Dr. Wright has something that the pioneering scientists of the antibiotic golden age did not: new techniques, new knowledge and a team of brilliant young scientists. And, perhaps most valuable of all, the fortitude – and faith – for placing a lifetime bet.
Salmonella sp. GDW, the strain of salmonella that infected Gerry D. Wright more than a decade ago, is kept and grown in the lab for study… and revenge.Blake Dillon/McMaster University/Supplied
In January, when a historic snowstorm buried southwestern Ontario, a bat signal was sent out on the Wright Lab’s Slack channel. Gerry’s salmonella was in trouble.
Dr. Wright had kept the pathogen that sickened him, holding it captive inside a vial labelled with his initials. The sample – affectionately referred to as “Gerry’s salmonella” – is now stored inside one of his lab’s many deep freezers alongside roughly 1,000 other drug-resistant pathogens, as well as Dr. Wright’s collection of more than 13,000 fungi and bacteria collected from around the world.
During the snowstorm, one of the freezers went out. “It was a mad scramble,” Dr. Wright said, adding that some lab members braved the weather to rescue the strains. “We don’t want them to thaw; a lot of them would die. These are the strains I’ve been collecting since I started doing this work back in the day.”
Dr. Wright’s sprawling collection of micro-organisms forms the invaluable backbone of his research. His hope is that it will some day yield an effective new defence against the looming crisis of antimicrobial resistance.
Growing up in North Bay, Ont., Dr. Wright was a kid who fantasized about playing drums for a rock band, not messing around with dirt in a lab. But he wound up studying chemistry at the University of Waterloo – one of the first in his family to pursue a university education. (He does occasionally moonlight as a drummer for a cover band called Null Hypothesis).
As a PhD student, it was another urgent public health crisis that captured his attention: HIV/AIDS. He was stunned to learn about these immunocompromised patients, many as young as he was, dying from strange fungal infections that should have been treatable.
“That had a big impact,” he said. “I really wanted to roll up my sleeves and do something.”
Around that time, a superbug was also starting to emerge. Doctors around the world were reporting cases of Enterococci bacteria that couldn’t be treated with vancomycin, an older antibiotic that had been discovered three decades earlier in the jungles of Borneo.
In 1990, Dr. Wright landed a post-doctoral position at Harvard Medical School, where he planned to study the enzymes involved in making bacterial cell walls. But just before arriving in Boston, his supervisor sent a note: Would he mind switching his post-doc project to vancomycin resistance instead?
Postdoctoral fellow Xuefei Chen inspects plates of fungal pathogens from the incubator.
“I rushed to the library to look up vancomycin,” he said. “I had no idea what it was.”
This was Dr. Wright’s introduction to antimicrobial resistance, a phenomenon that occurs when disease-causing bacteria, fungi or other microbes evolve to the point where drugs can no longer defeat them.
Bacteria have an astonishing array of strategies for resisting antibiotics, an arsenal developed over billions of years. But ever since the discovery of penicillin, humans have become gluttons for antibiotics – and their overuse in both health care and agriculture have supercharged an evolutionary process that favours the fittest microbes with the best defences.
Dr. Wright spent two years at Harvard studying the antibiotic resistance genes that can help bacteria overcome vancomycin. The work was fascinating; he had caught the AMR research bug.
When he was hired by McMaster University in 1993, he was determined to continue working on AMR, even though colleagues tried to warn him off. “You’re never, ever, ever going to get a dime to work in this field,” he recalled being told. “I kind of stuck to my gut.”
At first, he focused on trying to build or find molecules that can block the mechanisms bacteria have for resisting antibiotics. But Dr. Wright couldn’t shake his fascination with vancomycin resistance.
Like most antibiotics, vancomycin is produced naturally by a soil bacterium. Scientists still don’t fully understand the role of antibiotics in the natural environment but one theory is they’re used as chemical weapons against rival microbes.
Dr. Wright wanted to know how bacteria make vancomycin while also avoiding accidental self-destruction. He decided to sequence its genome, a technique that was still relatively new and difficult in the late 1990s. Once he unfurled its DNA, he spotted an old foe: the very same resistance genes that he’d spent two years studying at Harvard.
A lightbulb went off. Scientists had spent decades trying to understand where antibiotic resistance was coming from, but investigations had mostly fixated on the pathogens that show up in patients. Could the source of all the resistance be the soil bacteria that make these antibiotics in the first place, he wondered?
“That’s when it dawned on me,” he said. “If you really want to understand the evolution of resistance, you should go and find it in the wild.”

With his tortoiseshell glasses and white lab coat, Dr. Wright looks every bit a scientist. But he also considers himself to be something of an explorer – one who is not searching the distant stars or oceanic horizons, but the tiny galaxies of microbes that form an unseen film over our visible world.
“There are no more continents to be found,” he said. “But when you’re working in a biochemical lab, you’re constantly discovering new things. And that, to me, was just absolutely thrilling.”
After he found the vancomycin resistance genes, he wanted to know what other soil microbes were hiding in their genomes. He devised a plan: Collect as much soil as possible, isolate the microbes growing inside of it, and find the resistance genes they were harbouring.
As a research proposal, it was an impossible sell. “I could not get funding for this,” Dr. Wright said. “It was all done on fumes.”

Dr. Wright couldn’t get funding to collect soil samples for his research so most of the collection work was done by him, his lab members, and anyone else willing to lend a hand.Kevin Patrick Robbins/McMaster University/Supplied
He enlisted students from his lab, neighbours – even his own kids to help with sample collection. Whenever his young family went on hikes, he would arm his two children with a spoon and plastic baggie, directing them to run off and find some “interesting dirt.”
One summer, a student whose father worked for the Bank of Montreal was recruited to the cause. He sent e-mails to BMO branches in every “hamlet, province and territory,” according to Dr. Wright.
“It said, well, if you’re out somewhere, send a little bit of dirt to Gerry,” he recalled. “That brought us, you know, a couple hundred samples.”
Dr. Wright and his team wound up collecting 480 strains of bacteria, which they tested against 21 antibiotics. The results were stunning: Every single strain was resistant. Not just to a single antibiotic, but several. Two strains were resistant to 15.
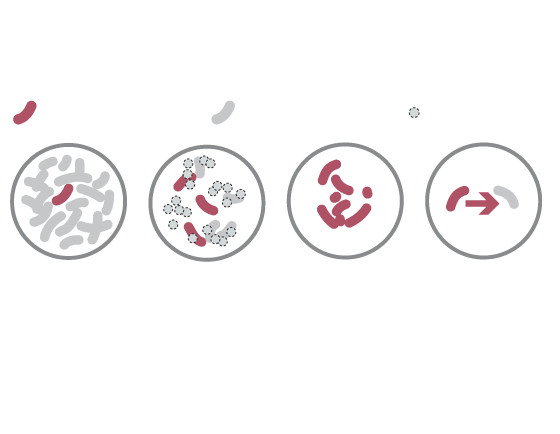
Rise of the superbugs
How antibiotic-resistant bacteria evolve
WHAT THEY ARE
Drug-resistant bacteria
Non-resistant bacteria
Dead bacteria
These drug- resistant bacteria can then grow and take over.
Usually, only
some bacteria are naturally resistant to drugs.
But when antibiotics kill non-resistant bacteria …
They can transfer drug-resistance to other bacteria.
Sources: reuters; Centers for Disease Control and Prevention; Antimicrobial Resistance Fighter Coalition
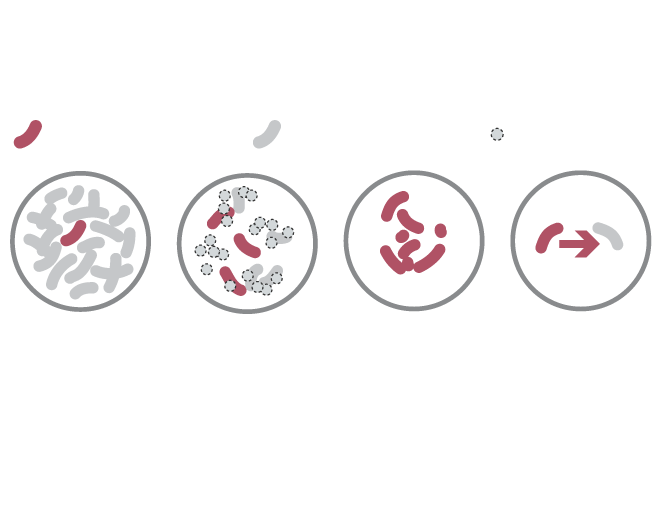
Rise of the superbugs
How antibiotic-resistant bacteria evolve
WHAT THEY ARE
Drug-resistant bacteria
Non-resistant bacteria
Dead bacteria
Usually, only
some bacteria are naturally resistant to drugs.
But when antibiotics kill non-resistant bacteria …
These drug- resistant bacteria can then grow and take over.
They can transfer drug-resistance to other bacteria.
Sources: reuters; Centers for Disease Control and Prevention; Antimicrobial Resistance Fighter Coalition
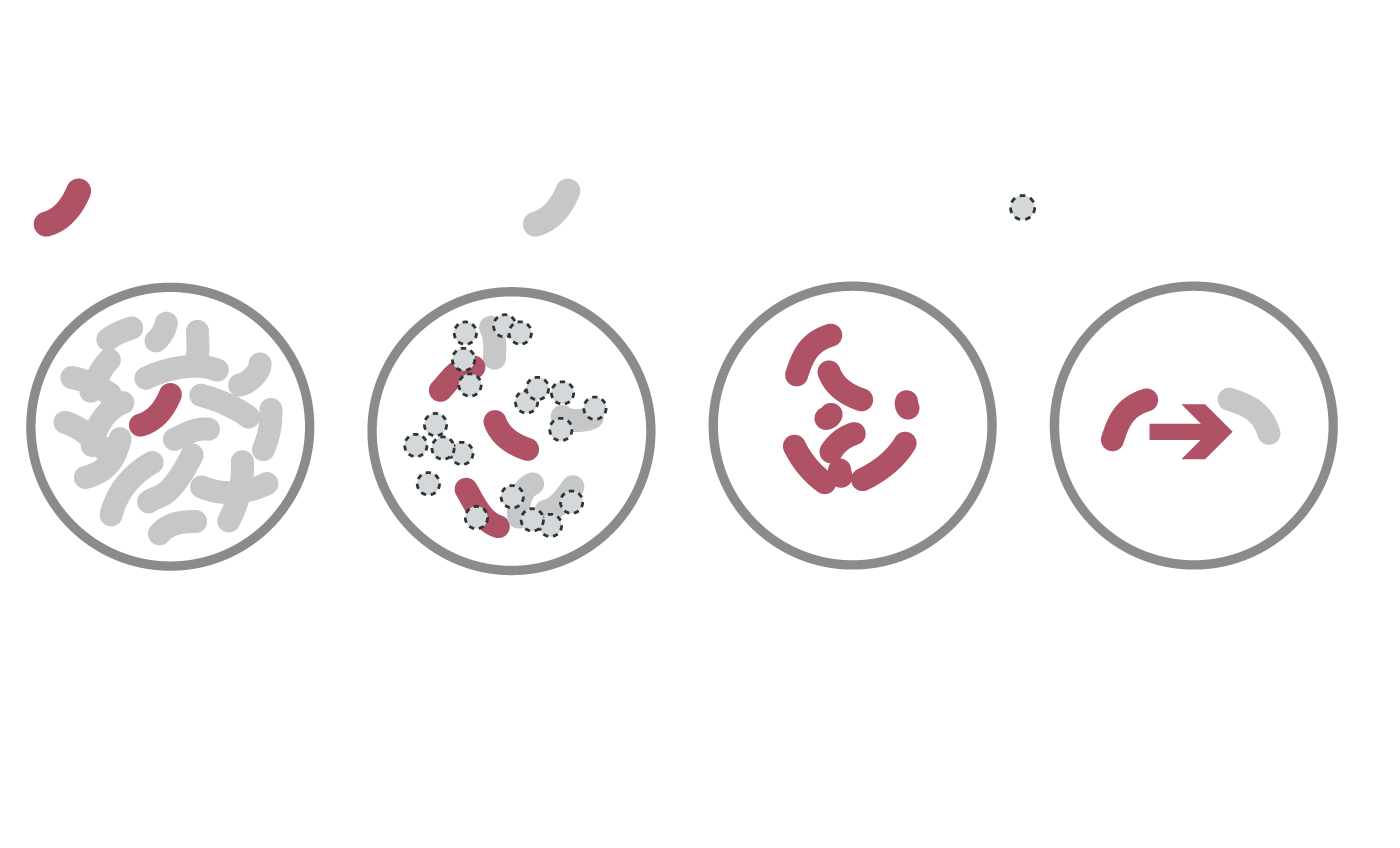
Rise of the superbugs
How antibiotic-resistant bacteria evolve
WHAT THEY ARE
Drug-resistant bacteria
Non-resistant bacteria
Dead bacteria
Usually, only
some bacteria are
naturally resistant
to drugs.
But when antibiotics kill non-resistant bacteria …
These drug- resistant bacteria can then grow and take over.
They can transfer drug-resistance to other bacteria.
Sources: reuters; Centers for Disease Control and Prevention; Antimicrobial Resistance Fighter Coalition
The 2006 study, published in the prestigious journal Science, landed with a splash. And it introduced a crucial concept into the scientific literature: the “resistome,” which refers to the totality of antibiotic resistance genes that circulate in the bacterial world.
“This concept has provided exceptional insights into the origin and dissemination of ARGs” wrote a 2021 review paper in the journal Experimental and Molecular Medicine, using the acronym for antibiotic resistance genes.
Scientists now understand the environmental resistome to be the origin and reservoir for genes that help superbugs overcome antibiotics. Among the many remarkable strategies bacteria have for acquiring resistance is “horizontal gene transfer” – the ability to share genes, even across different species.
“There’s a lot of genetic exchange between even vastly different bacteria – as evolutionary distant as, say, people and plants,” Dr. Wright said. “Bacteria love swapping DNA, and they’ve been doing it for millions and millions of years.”
Resistome research has revealed just how ancient these resistance genes are. In a 2011 paper, Dr. Wright and his team reported the discovery of some in bacteria that lived 30,000 years ago, long before the advent of medicinal antibiotics.

Dr. Wright’s research was illuminating the astonishing arsenal of tools that bacteria have for resisting antibiotics – even those that haven’t been discovered yet.
For someone working in antibiotic drug discovery, this might feel like a deflating message. But Dr. Wright saw an opportunity.
Because as he was peering into the genomes of soil bacteria, he noticed something else. Many of the bacteria known to produce antibiotics had the genetic programs to make dozens more. Perhaps, he thought, modern-day lab techniques could turn them on and coax these bacteria into producing their hidden antibiotics.
“These bacteria, they kind of hold their secrets tightly,” Dr. Wright said. “We had only seen the tip of the iceberg.”
The automated compound storage unit houses the lab's colossal trove of samples.
Dr. Wright had followed his hunch that studying soil bacteria would unearth fresh insights into antimicrobial resistance, and the gamble had paid off. But now he was left with a massive trove of samples, which he calls his “collection of brown goop.”
The brown goop, he decided, would be his next big bet. “We just spent five years collecting all these strains,” he said. “Maybe they actually produce something useful.”
There was once a time when most of the major pharmaceutical companies had their own brown goop collections that would have dwarfed Dr. Wright’s. After the discovery of penicillin, drug makers were motivated to find more “miracle drugs,” especially those capable of killing gram-negative bacteria – bacteria, like the ones that cause everything from whooping cough to cholera, that are enveloped by an extra membrane, making them particularly difficult to kill.
Penicillin was accidentally discovered inside a lab, but the vast majority of the world’s antibiotics have been pulled from soil bacteria – primarily actinomycetes. These bacteria, responsible for more than half of the antibiotics we use today, grow everywhere from the sea to the mountaintops and perform a critical role in breaking down organic matter in the soil. (Anyone familiar with petrichor, the pleasantly earthy aroma that follows a light rain, will have inhaled the aromatic compounds produced by actinomycetes.)
The microbe that produced streptomyces (seen here) belongs to a group of filamentous bacteria called actinomycetes, which can be found everywhere from the sea to the mountaintops.
The first medicinal antibiotic from an actinomycete was discovered in 1943, by researchers with Rutgers University in New Jersey who isolated the compound from bacteria growing inside the throat of a chicken and a handful of farm soil.
Medicines from actinomycetes have since been discovered on a Japanese golf course (the anti-parasitic ivermectin), a coal mine (a cancer drug) and Mount Ararat in Turkey, where scientists from the drug company Eli Lilly found daptomycin, an antibiotic that is now used as a last-resort treatment for superbugs such as methicillin-resistant Staphylococcus aureus, better known as MRSA.
But daptomycin was the last time anyone discovered a unique class of antibiotic that was successfully developed into a broadly useful medicine. And it was found in 1987, more than three decades ago.
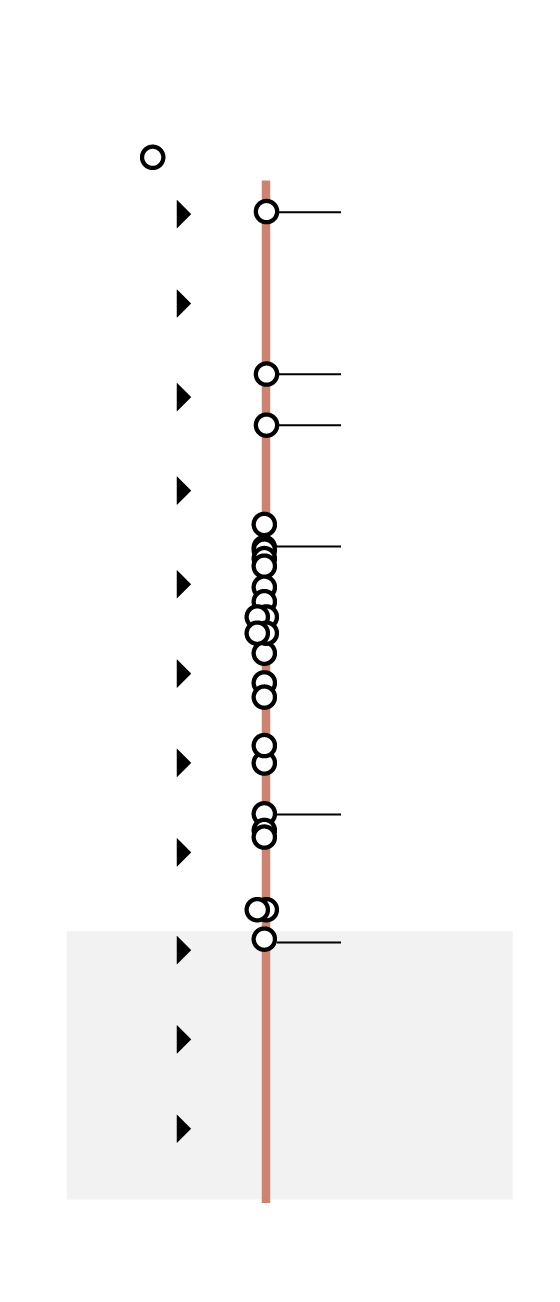
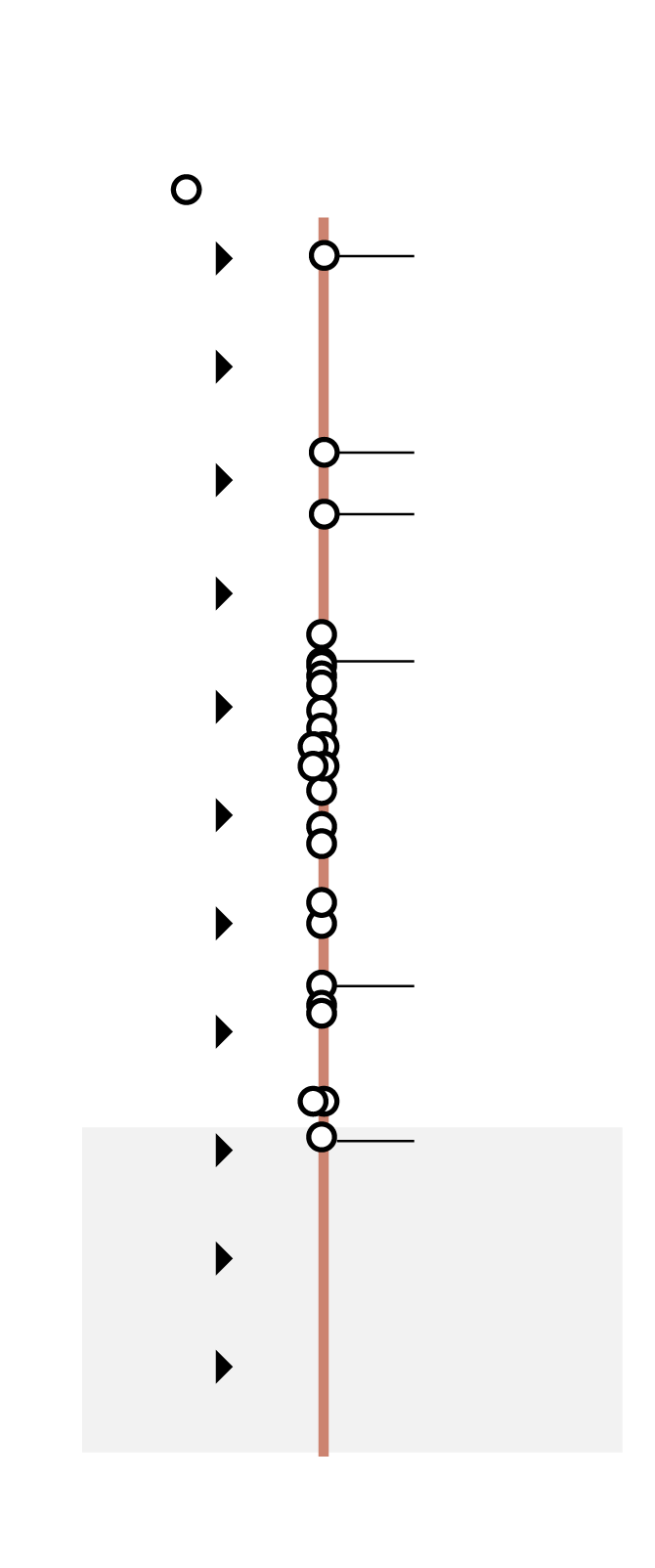
Timeline of antibiotic discovery
The discovery of daptomycin in 1987 was the last time
anyone found a unique class of antibiotic that was
successfully developed into a broadly useful medicine.
Major drug class
1910
Salvarsan
1920
Penicillin
1930
Sulphonamides
1940
Tetracyclines
1950
1960
1970
Carbapenems
1980
Daptomycin
1990
2000
Discovery
void
2010
the globe and mail, Source: nature
Timeline of antibiotic discovery
The discovery of daptomycin in 1987 was the last time
anyone found a unique class of antibiotic that was
successfully developed into a broadly useful medicine.
Major drug class
1910
Salvarsan
1920
Penicillin
1930
Sulphonamides
1940
Tetracyclines
1950
1960
1970
Carbapenems
1980
Daptomycin
1990
2000
Discovery
void
2010
the globe and mail, Source: nature
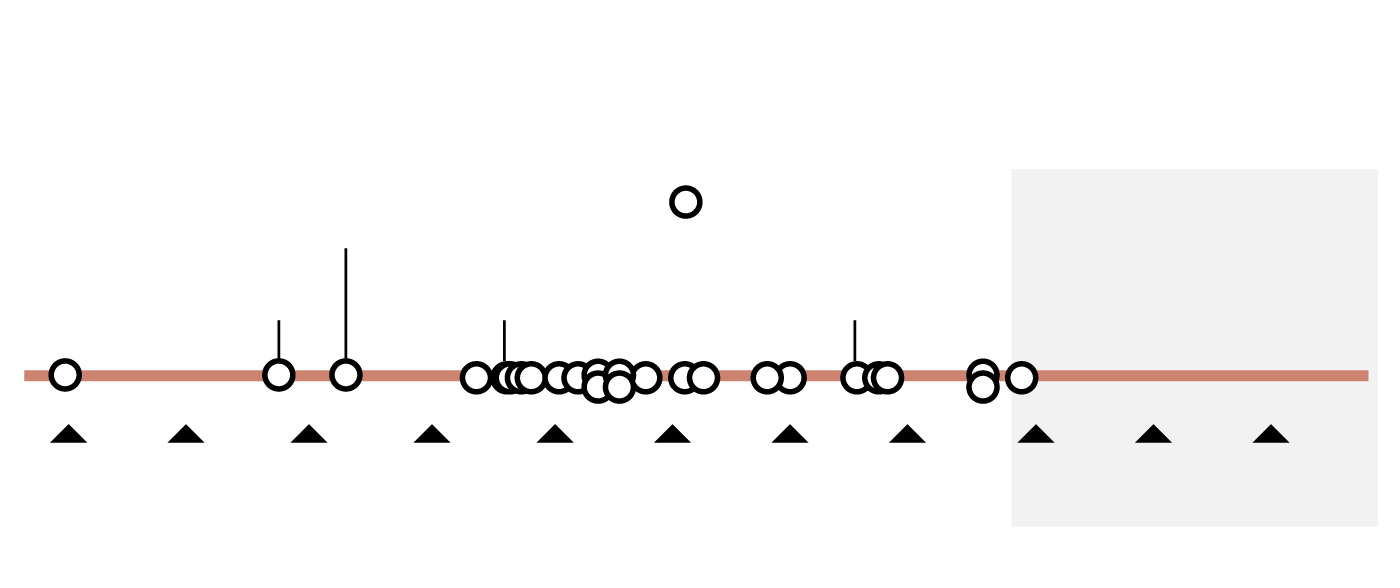
Timeline of antibiotic discovery
The discovery of daptomycin in 1987 was the last time anyone found a unique class of antibiotic
that was successfully developed into a broadly useful medicine.
Major drug class
Sulphonamides
Discovery void
Penicillin
Tetracyclines
Carbapenems
Daptomycin
Salvarsan
1910
1920
1930
1940
1950
1960
1970
1980
1990
2000
2010
the globe and mail, Source: nature
As drug companies stopped finding antibacterial compounds – and as pathogenic bacteria became increasingly resistant – the antibiotics market began to collapse. Big Pharma started shifting its investments toward more lucrative diseases, such as cancer.
The work of antibiotic discovery is now largely left to small biotechs and academic labs. Most of the remaining AMR researchers are placing their chips on novel approaches – searching extreme environments, such as deserts and ancient tundra; using artificial intelligence; or working with bacteriophages, which are viruses that infect bacteria.
Only a few are betting on actinomycetes. Dr. Lewis at Northeastern University, who runs one of the world’s top academic labs in the field, prefers to focus on soil’s “dark matter” – the 99 per cent of bacteria that have never been tested because they’ve historically been impossible to culture in the lab.
“My take is very simple: Let’s screen outside of actinomycetes,” said Dr. Lewis, who describes Dr. Wright as one of the world’s top three experts in natural antibiotic discovery. “Gerry says I’m wrong … and I sincerely hope that he’s right.”
Dr. Wright, a son of northern Ontario, uses a mining analogy to explain his rationale for going all in on soil bacteria such as actinomycetes. The antibiotic hunters of yesteryear were merely chipping away at the surface of the antibiotic gold mine, he says. Today’s scientists have far more powerful tools – and a dramatically better understanding of the microbial world – enabling them to dig deeper into the mine, where he believes more treasures can be found.
“If you think of the antibiotic discovery over the last 80 years, the first part was the easy ore to get, right?” he said. “But now, with new techniques and new technology, you go back to that slag – and you can pull out another fortune.”
The Wright Lab is filled with a diverse team of young scientists from around the world, such as postdoctoral fellow Manoj Jangra from India.Nick Iwanyshyn/The Globe and Mail
But no microbial miner can go it alone, especially not with a gold mine as massive as Dr. Wright’s library of brown goop – and the estimated 100,000 compounds it produces, only about 5 per cent of which have been screened so far.
Dr. Wright knows he needs help from people with perspectives and expertise that he lacks. At the Wright Lab, where boxes are crammed on every shelf and beakers crowd the countertops, the sunlit space features a diverse team of young scientists from around the world, including Sweden, China and India.
They are molecular biologists, geneticists and experts in bioinformatics. And they have expertise in everything from genetics and synthetic biology to enzyme studies and protein structures.
“Normally labs do just one of those things, or two. It’s odd,” Dr. Wright said of the mix. “But I try to bring in people with different skill sets.”
This breadth of knowledge was key in attracting Manoj Jangra and his wife, Manpreet Kaur, all the way from Chandigarh, India.
As budding chemical biologists, they had idolized Dr. Wright from afar and dreamed of working with him. So when they learned he was speaking at a conference in a nearby town in 2019, they knew they had to attend.

Dr. Gerry Wright, Dr. Manoj Jangra and Eric Brown, professor of biochemistry and biomedical sciences at McMaster, and one of Gerry's longest running collaborators at the conference where Dr. Jangra met Dr. Wright in 2019 in India.Manoj Jangra/Supplied
It felt like meeting a celebrity, Dr. Jangra said. “We had a couple of photos with Gerry. That was the best part.”
Dr. Kaur, then his girlfriend, managed to secure a post-doctoral fellowship in the Wright Lab. Once she was in Hamilton, she started nudging her boss to bring her talented “friend” over from India.
“She tried a lot. She worked day and night; even during COVID, she was here,” Dr. Jangra said. “Just so she can impress him and she can convince him to have a talk or interview with me.”
In academia, it’s “impossible” for couples to get post-doc positions at the same prestigious lab, Dr. Jangra said. But once Dr. Wright learned the two researchers were an item, he felt he had to reunite them.
“They were … separated in two different continents,” Dr. Wright recalled. “I said, all right, I can afford this. I can make sure that they’re together.”
In 2021, Dr. Jangra joined Dr. Kaur at the lab, and two months later they were married. Dr. Wright walked Dr. Kaur down the aisle, and the entire team attended the celebrations. In June, the pair welcomed their first child.
Shortly after arriving, Dr. Kaur pitched an unorthodox idea. One of the lab technicians had gifted her a bag of backyard soil, and she’d managed to isolate more than 300 microbes in Petri dishes. She wanted to starve them of nutrients – and then wait for a year.
“That’s an experiment that I never would have done,” Dr. Wright said. “But I figured, knock yourself out, it’s an interesting idea. I didn’t have a great reason for thinking it wasn’t going to work.”
When the 12 months were up, Dr. Jangra, who had arrived in Canada, joined the effort. He homed in on an antibiotic compound produced by a Paenibacillus bacterium from one of his wife’s Petri dishes.
Over the next three years, he conducted a series of experiments confirming that not only had this antibiotic never been discovered before, it represented a novel class – the holy grail in antibiotic drug discovery, because it means that current superbugs won’t have pre-existing resistance.
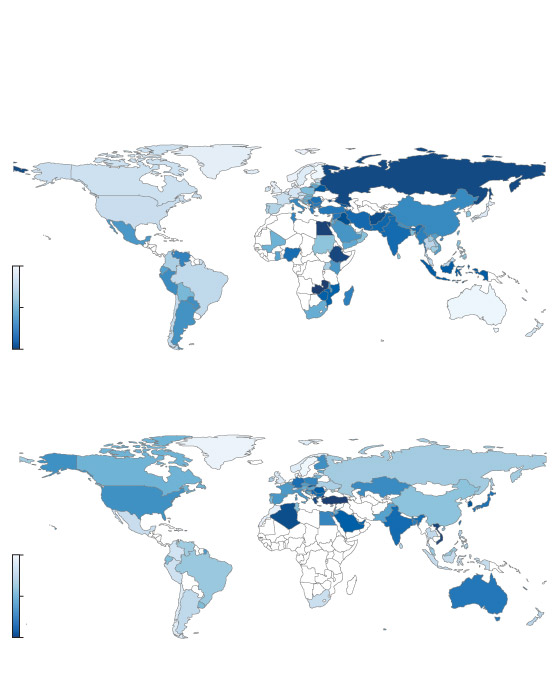
A worldwide problem
Critical pathogen antibiotic resistance rates and consumption
of carbapenem and cephalosporin (broad-spectrum antibiotics),
by country
Critical pathogen antibiotic resistance rates
Minimum 5.6%
Median 36.9%
Maximum 94.0%
Carbapenem and cephalosporin consumption
In defined daily doses per 1,000 individuals
Minimum 44.7
Median 540.1
Maximum 2,658.9
the globe and mail, Source: the lancet, april 2023
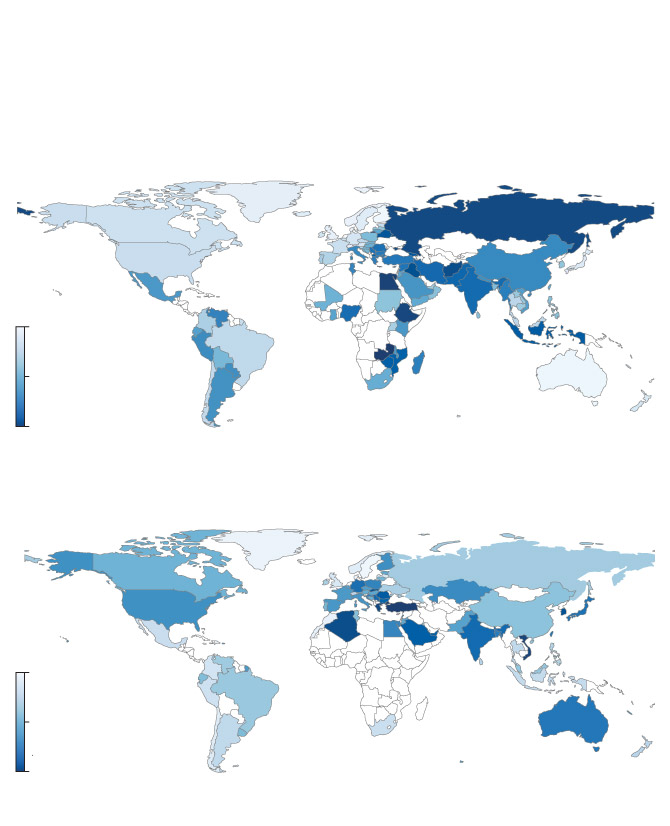
A worldwide problem
Critical pathogen antibiotic resistance rates and consumption
of carbapenem and cephalosporin (broad-spectrum antibiotics),
by country
Critical pathogen antibiotic resistance rates
Minimum 5.6%
Median 36.9%
Maximum 94.0%
Carbapenem and cephalosporin consumption
In defined daily doses per 1,000 individuals
Minimum 44.7
Median 540.1
Maximum 2,658.9
the globe and mail, Source: the lancet, april 2023
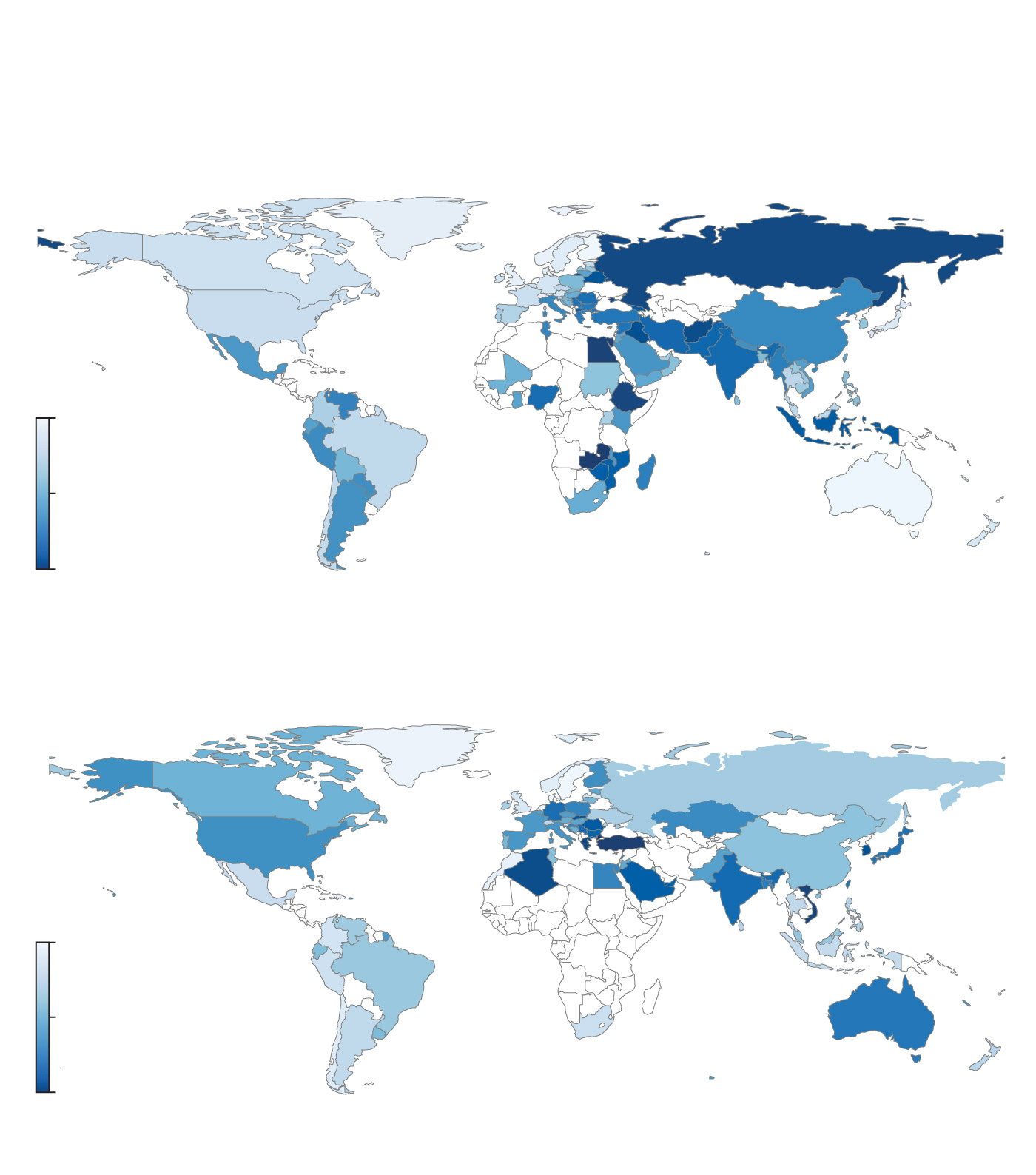
A worldwide problem
Critical pathogen antibiotic resistance rates and consumption of carbapenem and cephalosporin
(broad-spectrum antibiotics), by country
Critical pathogen antibiotic resistance rates
Minimum 5.6%
Median 36.9%
Maximum 94.0%
Carbapenem and cephalosporin consumption
In defined daily doses per 1,000 individuals
Minimum 44.7
Median 540.1
Maximum 2,658.9
the globe and mail, Source: the lancet, april 2023
When they tested the antibiotic against drug-resistant pathogens – including Gerry’s salmonella – they discovered that it successfully treated mice infected with one of the world’s most worrisome superbugs.
They named their discovery lariocidin, after its lariat-like structure, and published a paper in Nature last March. Lariocidin exhibits qualities that are “currently extremely rare in the search for new antibiotics,” wrote a commentary published by the renowned journal. Such a finding, it noted, “should rightly be met with great fanfare.”
A 3D-printed model of lariocidin, a new antibiotic molecule recently discovered in the Wright Lab.
But lariocidin was just one of three novel antimicrobial discoveries published by the Wright Lab this year, along with an adjuvant – or helper molecule, which can revive the efficacy of existing antibiotics – and an anti-fungal, which was found thanks to the dogged persistence of Xuefei Chen, another post-doctoral fellow. They have two more undergoing peer review, including a second novel antibiotic from Dr. Kaur’s Petri dishes – this one made by an actinomycete.
For Dr. Chen, a key ingredient in the lab’s success has been a work environment that values collaboration and scientific freedom while also putting its people first. She was recently hired as an associate professor at University of Oxford, where she will be starting her own lab and “immigrating” the Wright Lab culture.
“When I first came here, Gerry always encouraged me,” she said. “He was like, Fei, let’s save the world.”
After three decades, thousands of soil samples and hundreds of scientific papers, Dr. Wright is closer than ever to finding a new bulwark against the growing tide of antimicrobial resistance.
But everything that came before was, arguably, the easy part. Dr. Wright now faces his toughest challenge yet: getting his antimicrobials into medicine cabinets.
These days, he spends most of his time trying to persuade someone, anyone, to invest the money he needs to develop lariocidin into a drug. So far, he’s failed to secure any funding from North American investors or grants.
Imagine, for a moment, that you’re a pharmaceutical company. Now here comes the pitch for investing in a new antibiotic:
This is a drug that will probably take a decade and more than US$1-billion to develop, while generating annual revenues that are only a fraction of that amount. Patients will only need it for days or weeks, and doctors will be reluctant to prescribe it. The laws of Darwinism all but guarantee your drug will eventually stop working as bacteria develop resistance.
And there’s always a risk you’ll go under trying to bring this drug to market, as was the case for South San Francisco-based company Achaogen, which filed for bankruptcy less than a year after the U.S. Food and Drug Administration approved an antibiotic it was developing.
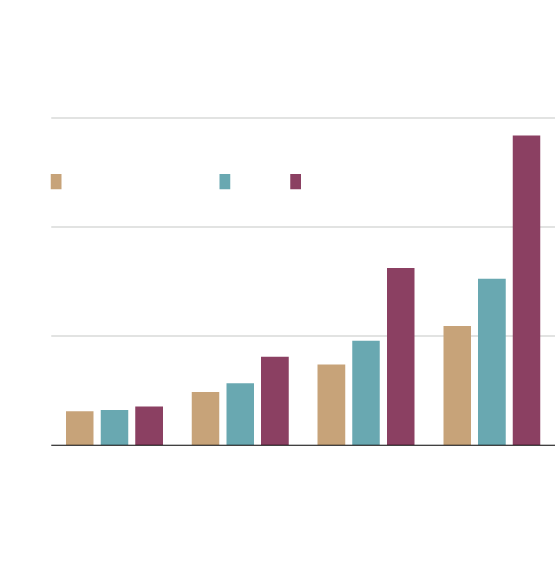
Projected Canadian annual hospital costs
associated with antimicrobial resistance
In billions of dollars, 2020-2050
$15
AMR accounts for
0.9 to 1.6% of
health spending
Resistance rate
26% (status quo)
40%
100%
10
5
AMR accounts for
0.6% of health
spending
0
2020
2030
2040
2050
In the Panel’s model: status quo (26%), 40%, and 100%. It assumes that hospital costs
grow at 4.2% per year from 2020 to 2050.
the globe and mail, Source: Council of Canadian Academies
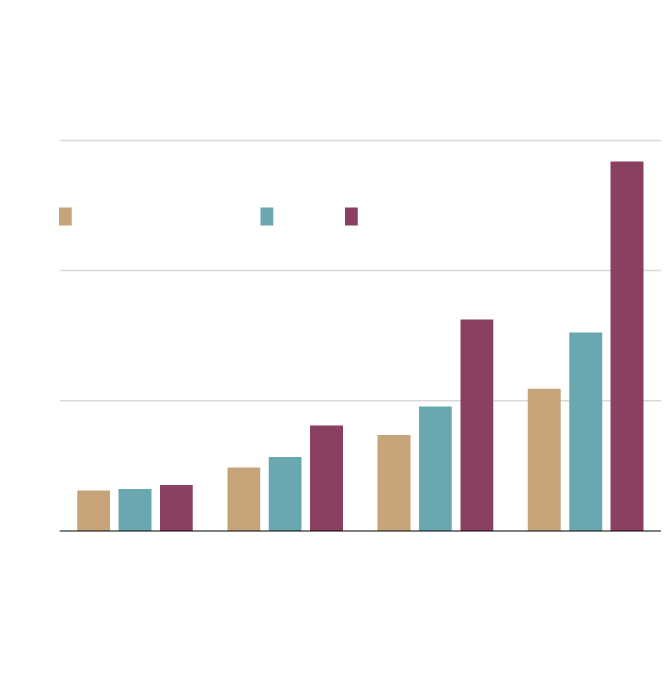
Projected Canadian annual hospital costs
associated with antimicrobial resistance
In billions of dollars, 2020-2050
$15
AMR accounts for
0.9 to 1.6% of
health spending
Resistance rate
26% (status quo)
40%
100%
10
5
AMR accounts for
0.6% of health
spending
0
2020
2030
2040
2050
In the Panel’s model: status quo (26%), 40%, and 100%. It assumes that hospital costs
grow at 4.2% per year from 2020 to 2050.
the globe and mail, Source: Council of Canadian Academies
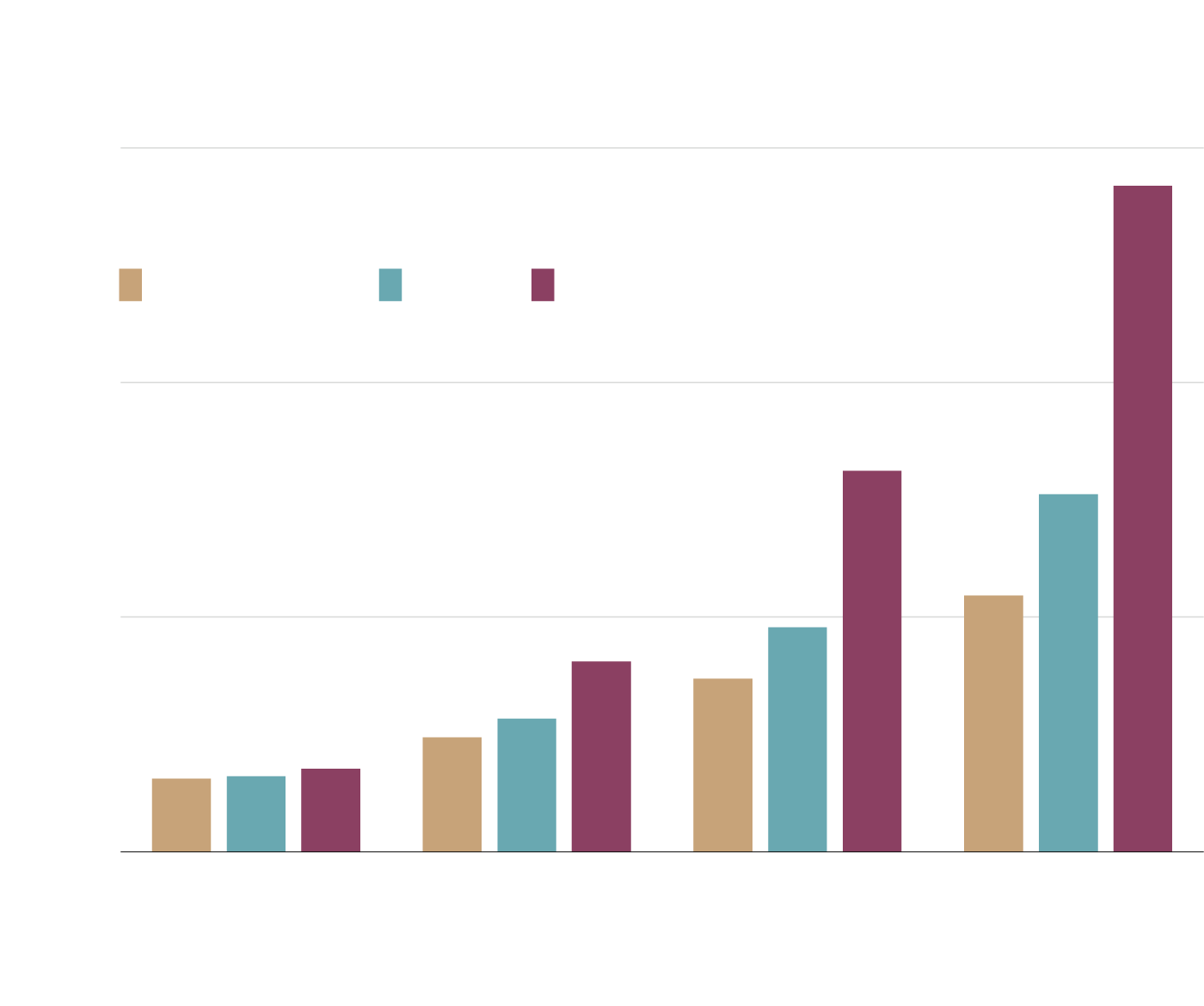
Projected Canadian annual hospital costs associated with antimicrobial resistance
In billions of dollars, 2020-2050
$15
AMR accounts for
0.9 to 1.6% of
health spending
Resistance rate
26% (status quo)
40%
100%
10
5
AMR accounts for
0.6% of health
spending
0
2020
2030
2040
2050
In the Panel’s model: status quo (26%), 40%, and 100%. It assumes that hospital costs grow at 4.2% per year from 2020 to 2050.
the globe and mail, Source: Council of Canadian Academies
Meanwhile, drugs for other diseases, such as cancer, can make you three or even 11 times more money.
It’s easy to see why this is a losing argument, even for pharmaceutical executives who care about antimicrobial resistance.
“You are working in a desert,” Philippe Villain-Guillot, former chief executive officer of the drug company Nosopharm, told Politico last year about his efforts to sustain an antibiotic program. “Unfortunately, the market is broken for antibiotics.”
Dr. Wright knows from personal experience just how quickly a promising antibiotic can perish in the “valley of death” of drug development.
On his office wall, he’s hung a framed cover of the June, 2016, issue of Nature, which featured another promising compound he discovered more than a decade ago. Dr. Wright even founded a company to further develop it.
But two animals died during preclinical tests, for reasons that were never understood. Overnight, the entire operation was shut down.
“There are a million ways this can derail,” he said.
But at an age when many people start leaning into their retirements, Dr. Wright is back to being a full-time scientist, having recently stepped down from many of his administrative positions, including as the inaugural director of McMaster’s infectious disease research institute.
He has spent more than half of his life chipping away at the colossal problem of antimicrobial resistance. And he’s finally hitting his professional stride.
“The thing about science … it’s not for the risk averse,” he said. “If you want to discover new things, you have to be willing to stick your neck out.
“It’s more than 25 years of building in this area to get to here. Which is why I kind of want to keep going.”
