Sanofi Tops Q1 Earnings Estimates as Dupixent & New Drugs Drive Growth

Sanofi SNY reported first-quarter 2026 adjusted earnings of $1.10 per American depositary share, which beat the Zacks Consensus Estimate of $1.06 per share. Earnings of €1.88 per share rose 5% on a reported basis and 14.0% on a constant currency rate (“CER”) basis, driven by cost control.
Net sales rose 6.2% on a reported basis to $12.3 billion (€10.51 billion). Sales rose 13.6% on a CER basis. Sales beat the Zacks Consensus Estimate of $11.99 billion.
Sales rose 26.2% at CER in the United States, 0.2% in the Rest of the World (including China, Japan, Brazil and Russia) and 5.9% in Europe.
All growth rates mentioned below are on a year-on-year basis and at CER.
SNY’s Dupixent Continues to Outperform
In Immunology, blockbuster drug Dupixent generated sales of €4.17 billion in the quarter, up 30.8% year over year, driven by strong prescription trends across all approved indications and geographies.
Sales of the drug in the United States rose 35.9%. Dupixent sales rose 22.4% in Europe and 14.9% in the Rest of the World.
Sanofi markets Dupixent in partnership with RegeneronREGN. While sales are recorded by Sanofi, Regeneron records its share of profits/losses in connection with global sales of Dupixent.
SNY’s Rare Disease Drugs’ Performance
Among Sanofi’s rare disease drugs, the rare blood disorder drug, Altuviiio, a once-weekly new class of factor VIII therapy for hemophilia A, recorded sales of €325 million in the first quarter, up 42.2% year over year, mainly driven by patient switches in the U.S. hemophilia A market and launch in Japan and Taiwan. As much as 83% of Altuviiio’s sales were in the United States.
Among the rare disease drugs in the Pompe franchise, Nexviazyme/Nexviadzyme recorded sales of €208 million, up 13.3% year over year, mainly driven by strong sales growth in Europe. Myozyme sales declined 13.3% year over year to €112 million due to patients switching to Nexviazyme. Fabrazyme sales were €265 million, up 7.6% year over year. In the Gaucher franchise, Cerezyme sales rose 1.1% year over year to €186 million.
Cablivi recorded sales of €68 million, up 9% year over year, driven by patient growth in the United States and Europe.
Among the rare blood disorder drugs, Eloctate sales declined 7.1% to €58 million in the quarter due to patients switching to the new drug Altuviiio.
Xenpozyme recorded sales of €63 million in the quarter, up 17.9% year over year.
New drug Qfitlia (fitusiran) generated €5 million in sales, compared with €4 million in the previous quarter.
Another new drug, Wayrilz (rilzabrutinib) generated €10 million in sales in the first quarter versus €6 million in the previous quarter.
Ayvakit, added from last year’s acquisition of Blueprint Medicines, recorded sales of €177 million compared with €168 million in the previous quarter, driven by continued patient growth. Higher government rebates hurt U.S. sales to an extent.
SNY’s Oncology & Other Drugs
In Oncology, Sarclisa sales rose 30.1% year over year to €167 million, driven by increased use in earlier lines of treatment in Europe and strong growth from Rest of World.
In Others, Toujeo recorded sales of €375.0 million, up 10.5% year over year, driven mainly by volume growth and market share gains in the United States and Europe. Lantus sales declined 0.7% to €419 million. Rezurock recorded sales of €133 million, up 11.5% year over year. Tzield sales were €14 million in the quarter, up 36.4% year over year, driven mainly by U.S. sales.
Plavix sales declined 3.3% to €224 million. Lovenox sales decreased 22.3% to €184 million.
Heplisav-B Added from Dynavax Deal Boosts SNY’s Vaccine Sales
Total vaccine sales rose 2.1% to €1.29 billion, mainly due to higher sales of Sanofi and partner AstraZeneca’s AZN respiratory syncytial virus or RSV antibody, Beyfortusand Polio/Pertussis/Hib (PPH) vaccines.
Sales of PPH vaccines rose 4.2% year over year to €664 million, gaining from the inclusion of the newly acquired adult hepatitis B vaccine, Heplisav-B, from the acquisition of Dynavax, which closed in February. Sales of meningitis, travel and other endemic vaccines declined 2.0% year over year to €278 million in the quarter. Sales of flu vaccines declined 4.1% year over year to €67 million.
AstraZeneca-partnered Beyfortus recorded sales of €284 million in the quarter, up 2.8% year over year. Gains from expanded geographical availability in Europe and Rest of the World were partially offset by softer sales in the United States due to a tough comparison from inventory increase in the year-ago quarter.
SNY Maintains 2026 Guidance
Sanofi maintained its previously issued financial outlook for 2026.
Sanofi expects sales to rise by a high single-digit percentage at CER. Sanofi expects earnings to grow faster than sales in 2026 at CER, before the impact of share repurchases.
Our Take on SNY’s Q1 Results
Sanofi’s first-quarter results were strong as it beat estimates for earnings as well as sales. Both sales and earnings rose in a double-digit range at CER in the quarter. Higher sales of Dupixent and contributions from new products like Altuviiio and Ayvakit drove top-line growth. Vaccine sales also improved in the quarter, benefiting from the inclusion of Heplisav-B. Dupixent continues to be the key top-line driver with global sales rising above €4 billion for the past three consecutive quarters.
Sanofi’s shares were up more than 4% in pre-market trading in response. Sanofi’s stock has declined 12.4% in the past year against the industry’s increase of 12.9%.
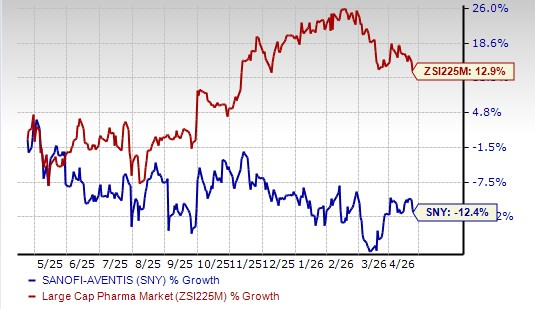
Image Source: Zacks Investment Research
Sanofi is seeing a good uptake of its new medicines and vaccines. Altuviiio achieved blockbuster sales in 2025 and Ayvakit is expected to become the next blockbuster drug in 2026. Last year, Sanofi launched Qfitlia and Wayrilz. Sales of Sanofi’s newly launched drugs and vaccines rose almost 50% in the first quarter.
Some potential blockbuster assets in phase III development are amlitelimab (anti-OX40L mAb for moderate-to-severe atopic dermatitis) and frexalimab (anti-CD40L mAb for multiple sclerosis). Regulatory applications for amlitelimab and frexalimab are planned in the second half of 2026 and 2027, respectively.
Sanofi has also been active on the M&A front. In February, it completed the previously announced acquisition of vaccine maker Dynavax Technologies for a total equity value of approximately $2.2 billion. Last year, it acquired Blueprint Medicines, which has expanded its presence in rare immunological diseases and Vigil Neuroscience, which expanded its early-stage pipeline in neurology. The company has also got a new chief executive officer, Belén Garijo, who will replace Paul Hudson, effective April 29, 2026.
SNY’s Zacks Rank
Sanofi currently has a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Zacks' Research Chief Names "Stock Most Likely to Double"
Our team of experts has just released the 5 stocks with the greatest probability of gaining +100% or more in the coming months. Of those 5, Director of Research Sheraz Mian highlights the one stock set to climb highest.
This top pick is a little-known satellite-based communications firm. Space is projected to become a trillion dollar industry, and this company's customer base is growing fast. Analysts have forecasted a major revenue breakout in 2025. Of course, all our elite picks aren't winners but this one could far surpass earlier Zacks' Stocks Set to Double like Hims & Hers Health, which shot up +209%.
Free: See Our Top Stock And 4 Runners UpThis article originally published on Zacks Investment Research (zacks.com).